Gut-Brain Axis: How Your Microbiome Affects Mood and Cognition
Your gut produces 90% of your body's serotonin and communicates with your brain through the vagus nerve. Learn what current research says about the microbiome-mood connection.
14 Min Read
TL;DR: Your gut houses over 500 million neurons, manufactures roughly 90% of your body's serotonin, and maintains constant two-way communication with your brain through the vagus nerve. Disruptions to this system are linked to anxiety, depression, and cognitive decline, and targeted dietary changes can measurably improve the balance.
Your Gut Contains a Second Nervous System
Buried in the walls of your gastrointestinal tract is a network so complex that neuroscientists call it the "second brain." The enteric nervous system (ENS) contains more than 500 million neurons, which is more than your spinal cord holds, and it stretches from your esophagus all the way to your rectum. Unlike any other organ system, the ENS can operate without instructions from your actual brain. It coordinates digestion, manages blood flow to the intestinal lining, and triggers immune responses on its own.
But the ENS does more than push food through your intestines. It manufactures over 30 different neurotransmitters, the same chemical messengers your brain uses to regulate mood, focus, and sleep. Dopamine, GABA, acetylcholine, and serotonin are all produced in substantial quantities right inside your gut wall. Your digestive tract also harbors about 70% of your body's immune cells and is home to over 100 trillion microorganisms, collectively known as your gut microbiome.
If you've ever dealt with anxiety or chronic stress, this matters more than you might think. The enteric nervous system doesn't just respond to what you eat. It responds to emotional states, hormonal shifts, and bacterial populations. When researchers at Washington University eliminated all gut bacteria from laboratory mice, the animals showed dramatically altered stress responses, with exaggerated cortisol output and measurably reduced levels of brain-derived neurotrophic factor (BDNF), a protein that's critical for learning and memory. Reintroducing specific bacterial strains reversed these changes, which confirmed that gut microbes are environmental determinants that regulate the stress response.
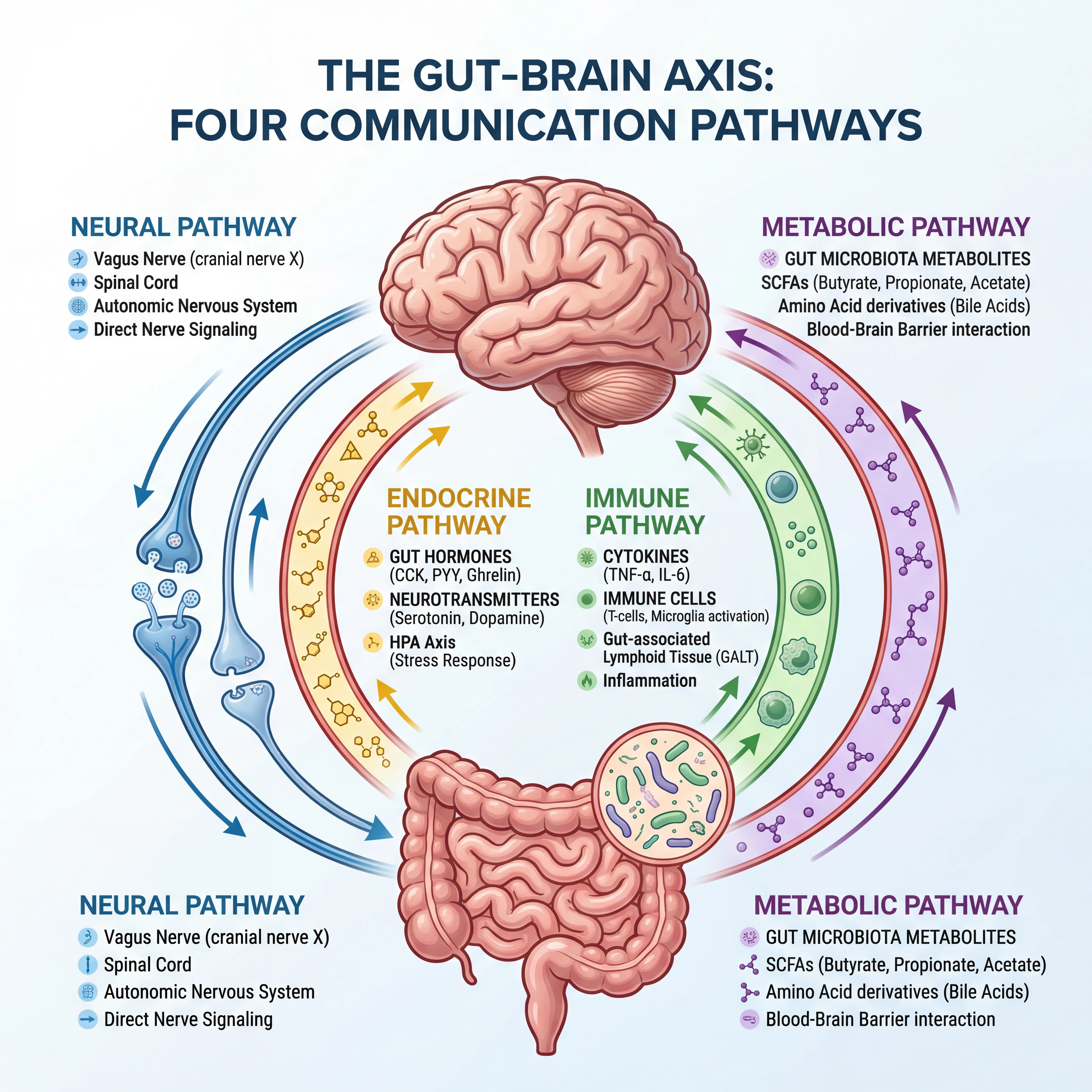
The gut-brain axis is bidirectional. Traffic flows both ways. Your brain can alter the composition of your gut bacteria through stress hormones and autonomic nerve signals that change intestinal motility, secretion, and immune function. Your gut bacteria, in turn, produce metabolites and neurotransmitters that travel back to the brain and influence how you think and feel. Some researchers have taken to calling this the "gut connectome" because of how many overlapping communication channels are involved.
The Vagus Nerve: A Direct Line Between Belly and Brain
If the gut-brain axis is a communication highway, the vagus nerve is the main cable. Named from the Latin word for "wanderer," the vagus is the longest cranial nerve in the body. It starts in the brainstem and threads its way through the neck, chest, and abdomen, branching into the heart, lungs, and every major section of the digestive tract.
Here's what most people don't realize: the vagus nerve is mostly a listening device. About 80 to 90% of its fibers are afferent, which means they carry information from the body up to the brain, not the other direction. Your gut is constantly feeding data about its chemical environment, bacterial populations, nutrient absorption, and inflammatory status directly into the brainstem's nucleus tractus solitarius (NTS). From there, signals get routed to the hypothalamus, amygdala, and prefrontal cortex, the regions responsible for appetite, fear, and decision-making.
| Vagus Nerve Function | Direction | What It Communicates |
|---|---|---|
| Sensory (afferent) | Gut → Brain | Nutrient status, bacterial metabolites, inflammation levels, mechanical stretch |
| Motor (efferent) | Brain → Gut | Digestive regulation, heart rate modulation, immune cell activation, anti-inflammatory signals |
The vagus nerve also has a built-in anti-inflammatory reflex. When vagal efferent fibers fire, they release acetylcholine, which binds to receptors on immune cells and suppresses the production of pro-inflammatory cytokines like TNF-alpha and IL-6. That's a big part of why vagus nerve stimulation (VNS) is being studied as a treatment for treatment-resistant depression, PTSD, and inflammatory bowel disease. The nerve isn't just relaying messages. It's actively tamping down the inflammatory processes that feed into psychiatric conditions.
Your "vagal tone," a measure of how actively your vagus nerve responds, tracks closely with your ability to regulate stress. People with higher vagal tone tend to recover faster from emotional disturbances. Slow diaphragmatic breathing, meditation, and cold water exposure have all been shown to increase vagal tone, which may partly explain why these practices seem to help with mood and resilience.
Where Most of Your Serotonin Actually Lives
If you think serotonin is mainly a brain chemical, the numbers say otherwise. Roughly 90% of the body's serotonin is made in the gut, mostly by specialized cells called enterochromaffin cells that line the intestinal wall. These cells monitor the chemical environment inside your intestines and release serotonin in response to food, bacterial metabolites, and mechanical stimulation.
Gut-derived serotonin doesn't cross the blood-brain barrier to become brain serotonin directly. Instead, it activates vagal afferent nerve endings locally, and those signals travel up through the vagus nerve to the brainstem. From the NTS, serotonergic signals reach the dorsal raphe nucleus, which is the brain's primary serotonin-producing region, and the locus coeruleus, which governs norepinephrine output. Through this relay system, conditions inside your intestines directly affect how much serotonin and norepinephrine your brain produces.
| Neurotransmitter | % Produced in Gut | Primary Gut Function | Primary Brain Function |
|---|---|---|---|
| Serotonin (5-HT) | ~90% | Gut motility, secretion | Mood, sleep, appetite |
| Dopamine | ~50% | Mucosal blood flow | Motivation, reward |
| GABA | Significant | Gut motility regulation | Anxiety reduction, calm |
The gut microbiome's role in serotonin production is well documented at this point. Bacteria produce short-chain fatty acids (SCFAs), primarily butyrate, propionate, and acetate, through the fermentation of dietary fiber. These SCFAs boost the expression of tryptophan hydroxylase 1 (TPH1), the rate-limiting enzyme in serotonin synthesis, essentially telling your enterochromaffin cells to ramp up production. SCFAs also affect the serotonin transporter (SERT), which controls how quickly serotonin gets cleared from the signaling environment.
SCFAs do more than just support serotonin. They can cross the blood-brain barrier and directly influence brain function. Research has shown that microbial SCFAs regulate microglia homeostasis, and microglia are the brain's resident immune cells responsible for synaptic pruning and neuroinflammation control. When SCFA production drops because of a fiber-poor diet or antibiotic-driven dysbiosis, microglial function deteriorates. That can contribute to cognitive fog and, over time, neurodegeneration. It's one reason dietary fiber keeps showing up in studies on brain health.
When Gut Bacteria Go Wrong: The Inflammation-Mood Connection
The lining of your intestine is just one cell layer thick, a fragile barrier separating the chaotic microbial world inside your gut from your sterile bloodstream. When this barrier weakens, a condition often called intestinal permeability or "leaky gut", bacterial components start crossing into circulation. The most studied of these trespassers is lipopolysaccharide (LPS), a molecule from the cell walls of gram-negative bacteria.
LPS in the bloodstream triggers a powerful immune response. Your body produces antibodies against it, and research has found that patients with major depression have significantly higher levels of anti-LPS antibodies than healthy controls. That's not a coincidence. It's one link in a chain connecting gut barrier dysfunction to brain inflammation.
The inflammatory cascade works through several mechanisms at once. When dysbiosis or barrier breakdown releases pro-inflammatory cytokines (IL-1-beta, IL-6, TNF-alpha), these molecules alter neurotransmitter metabolism by diverting tryptophan away from serotonin production and toward kynurenine, a neurotoxic pathway. They also activate the HPA axis to increase cortisol output and reduce BDNF expression in the hippocampus. All three of these effects are hallmarks of clinical depression.
| Gut Disruption | Inflammatory Mediator | Brain Effect |
|---|---|---|
| Dysbiosis (bacterial imbalance) | Elevated cytokines (IL-6, TNF-alpha) | Reduced serotonin synthesis, increased cortisol |
| Intestinal permeability | LPS translocation | Neuroinflammation, blood-brain barrier stress |
| Reduced SCFA production | Impaired microglial function | Cognitive fog, reduced BDNF |
| Chronic stress | Cortisol-driven gut changes | Anxiety, disrupted sleep |
And it goes both directions. Psychological chronic stress itself alters intestinal permeability. When the HPA axis stays activated for extended periods, cortisol reduces the expression of tight junction proteins that hold intestinal cells together. Stress-driven changes to autonomic nerve signaling also alter gut motility, secretion, and local immune function. The result is a feedback loop: stress damages the gut barrier, a damaged barrier fuels inflammation, inflammation worsens mood and cognition, and that produces more stress.
Parkinson's disease research has shown how deep this connection goes. GI dysfunction, particularly constipation and changes in gut bacterial composition, often appears years before the first motor symptoms. Some researchers now believe that alpha-synuclein aggregation may begin in the gut and travel to the brain via the vagus nerve. That makes the gut-brain axis relevant well beyond mood disorders.
What Clinical Trials Say About Probiotics and Mental Health
The idea that specific bacterial strains could improve mental health has moved well past animal research. A class of probiotics called "psychobiotics," bacteria specifically selected for their neurological effects, has been accumulating clinical evidence since the early 2000s. The most recent meta-analyses are cautiously positive.
A 2025 meta-analysis published in Brain and Behavior pooled data from randomized controlled trials and found that probiotic and prebiotic supplementation significantly reduced anxiety symptoms (standardized mean difference of 0.29 across 4,295 participants), significantly reduced depression symptoms (SMD of 0.29 across 3,179 participants), and significantly improved cognitive function (SMD of 0.48 across 915 participants). The cognitive improvement was the largest effect, nearly half a standard deviation.
To put those numbers in context: an SMD of 0.2 is considered a small effect in clinical research, and 0.5 is medium. The probiotic benefit for cognition approaches medium effect territory, which is comparable to some prescription interventions. The authors also found that people with milder symptoms tended to see greater benefits, which suggests probiotics may work better as preventive support than as a standalone treatment for severe conditions.

Earlier clinical trials helped pin down which strains show the most consistent effects. A triple-blind, placebo-controlled trial found that a multi-strain formula containing Bifidobacterium bifidum, B. lactis, Lactobacillus acidophilus, L. brevis, L. casei, L. salivarius, and Lactococcus lactis at 5 billion CFU/day significantly reduced cognitive reactivity to depression, specifically rumination and aggressive thoughts, in healthy young adults after just four weeks. Separate trials confirmed that combinations of L. helveticus and B. longum reduce depression, anger-hostility, and somatization scores. And a 2025 study in the International Journal of Molecular Sciences reported that L. acidophilus, B. bifidum, and Streptococcus thermophilus together significantly reduced the frequency of major depressive episodes after eight weeks of use.
The mechanism behind these effects probably involves multiple pathways working simultaneously. Beneficial bacteria strengthen the intestinal barrier, produce SCFAs that boost serotonin synthesis, affect vagal nerve signaling, and reduce the production of pro-inflammatory cytokines. No single mechanism explains the full effect, which makes sense given that the gut-brain axis itself operates through at least four parallel communication systems.
Myth vs. Fact: Common Misconceptions About the Gut-Brain Axis
| Myth | Fact |
|---|---|
| "Gut serotonin goes straight to the brain" | Gut serotonin can't cross the blood-brain barrier. It communicates with the brain indirectly by activating vagal afferents, which then modulate the dorsal raphe nucleus signal pathway. |
| "Any probiotic supplement will improve your mood" | Strain specificity matters. Only certain species and combinations (like L. helveticus, B. longum, and specific multi-strain formulas) have shown mental health benefits in controlled trials. A random yogurt brand won't necessarily help. |
| "The gut-brain axis is a new discovery" | William Beaumont experimentally showed that emotional states affect digestion in the 1840s. Hippocrates wrote about the connection between gut and brain health centuries before that. What's new is understanding the microbiome's role in it. |
| "You only need probiotics to fix gut-brain problems" | Probiotics work best alongside adequate dietary fiber (which feeds the bacteria), stress management (which protects the gut barrier), and reduced processed food intake. A single supplement can't compensate for a depleted ecosystem. |
| "The enteric nervous system is just a simple reflex network" | The ENS contains over 500 million neurons, produces 30+ neurotransmitters, and can operate independently from the brain. It is the most complex neural network outside the central nervous system. |
Feeding Your Microbiome for Better Mental Clarity
So what should you actually eat? The answer doesn't require exotic supplements or complicated meal plans. The core principle is bacterial diversity. The wider the range of beneficial species in your gut, the more robust your neurotransmitter production, SCFA output, and barrier integrity will be.
Dietary fiber is the single most important factor. Your gut bacteria ferment soluble and insoluble fiber into the SCFAs that drive serotonin synthesis, feed intestinal cells, and cross the blood-brain barrier to support microglial function. Most adults get around 15 grams of fiber per day, which is roughly half the recommended 25-30 grams. Increasing your intake from varied plant sources (legumes, whole grains, vegetables, fruits, nuts, seeds) can shift microbial composition within 24 hours.
Fermented foods provide live bacterial cultures. A Stanford University study found that participants who consumed six or more servings of fermented foods daily for 10 weeks showed significantly increased microbial diversity and reduced markers of inflammation. Practical options include yogurt with live cultures, kefir, sauerkraut, kimchi, miso, tempeh, and kombucha. The key is variety, since different fermented foods contain different bacterial strains.
Polyphenol-rich foods act as prebiotics. Dark berries, green tea, dark chocolate, and extra-virgin olive oil contain compounds that selectively promote the growth of beneficial bacteria like Bifidobacterium and Lactobacillus while inhibiting less desirable species. Those are the same genera that show up repeatedly in probiotic research on mood and cognition.

On the flip side: ultra-processed foods, artificial sweeteners, and emulsifiers (commonly found in packaged foods) have been shown to reduce microbial diversity and promote inflammatory bacterial strains. Excessive alcohol thins the intestinal mucus layer. And unnecessary antibiotic use can wipe out bacterial populations that take months to rebuild, which is one reason to use antibiotics only when medically necessary.
| Category | Foods to Emphasize | How They Help the Gut-Brain Axis |
|---|---|---|
| Fiber-rich | Beans, lentils, oats, artichokes, asparagus | Fuel SCFA production, feed beneficial bacteria |
| Fermented | Yogurt, kefir, kimchi, sauerkraut, miso | Introduce live beneficial cultures |
| Polyphenol-rich | Blueberries, green tea, dark chocolate, olive oil | Prebiotic effect, promote Bifidobacterium/Lactobacillus |
| Omega-3 sources | Fatty fish, walnuts, flaxseeds | Reduce neuroinflammation, support barrier integrity |
| Tryptophan sources | Turkey, eggs, cheese, nuts | Provide serotonin precursor |
Beyond diet, other lifestyle factors affect the gut-brain axis directly. Regular physical activity increases microbial diversity independently of diet. Sleep disruption alters bacterial composition within two days. Meditation and breathwork increase vagal tone. None of these work in isolation. Your microbiome responds to your entire pattern of living, not any single change.
Frequently Asked Questions
How long does it take for dietary changes to affect your gut microbiome?
Measurable shifts in bacterial composition can occur within 24 hours of a dietary change. Lasting changes to microbial diversity and abundance typically need 2-4 weeks of consistent effort, though. Adding fermented foods and high-fiber plants daily produces the most reliable long-term shifts.
Can probiotics replace antidepressant medication?
No. Current evidence supports probiotics as complementary support, not a replacement for prescribed psychiatric medications. The 2025 meta-analysis showed modest but significant effects on anxiety and depression, particularly for people with mild symptoms. Anyone on medication should talk to their doctor before making changes.
Does the gut-brain axis play a role in neurodegenerative diseases?
It appears to. In Parkinson's disease, gastrointestinal symptoms and altered gut bacteria often show up years before motor symptoms. Researchers believe misfolded proteins may travel from the gut to the brain via the vagus nerve. Similar connections are being studied in Alzheimer's disease and multiple sclerosis, though the evidence is still early.
What specific probiotic strains have the most evidence for mental health benefits?
Lactobacillus helveticus, Bifidobacterium longum, L. acidophilus, B. bifidum, and L. rhamnosus have the most clinical trial support. Multi-strain formulas containing several of these species at doses of 1-10 billion CFU daily have shown effects on depression, anxiety, and cognitive reactivity in randomized controlled trials.
Is the concept of "leaky gut" medically recognized?
Intestinal permeability is a well-documented physiological phenomenon studied extensively in gastroenterology. Stress, dysbiosis, certain medications, and dietary factors can increase the passage of bacterial components like LPS through the intestinal barrier. The pop-health term "leaky gut" sometimes attracts skepticism, but the underlying mechanism, impaired tight junction integrity, is accepted science with direct implications for systemic inflammation and mood.
Related Articles
- Leaky Gut Syndrome: Symptoms, Causes, and Evidence-Based Healing - A deep dive into intestinal permeability and how to restore your gut barrier.
- What Are the Best Probiotic Foods - A review of the top fermented foods and how they support digestive and immune health.
- The Hidden Toll of Low-Level Stress: How Chronic Tension Wrecks Your Hormones - How the HPA axis and cortisol affect gut health and overall wellbeing.
- Depression and Complementary Health Approaches - Evidence-based natural strategies for managing depressive symptoms.
- Collagen Supplements: Do They Actually Work for Skin, Joints, and Gut? - What the research says about collagen and digestive health.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.










