Peptides for Health: BPC-157, Thymosin Beta-4, and What's Legal
Evidence-based guide to BPC-157 and thymosin beta-4 peptides covering preclinical research, human clinical data, FDA classification, and current legal status.
12 Min Read
TL;DR: BPC-157 and thymosin beta-4 look promising in animal research, but human clinical data is thin. BPC-157 is an FDA Category 2 bulk drug substance, meaning pharmacies can't legally compound it. Neither peptide is FDA-approved, and the stuff sold online has no quality oversight.
Peptides have gone from biochemistry footnote to Reddit obsession. Scroll through any longevity podcast or fitness forum and you'll see BPC-157 and TB-500 discussed like they're creatine. They're not. The confidence people have in these compounds is way ahead of the clinical evidence supporting them.
That said, the preclinical data is genuinely interesting. The mechanisms these peptides activate, tissue repair, inflammation reduction, blood vessel formation, are real and well-documented in animal models. But "promising in rats" is a long way from "safe and effective in humans." About 90% of drugs that look good in animals fail to deliver in human trials. Where the science actually stands, and what the law says about buying these things, are two questions worth separating cleanly.
What peptides actually do in your body
Peptides are short amino acid chains, typically 2 to 50 residues long. Your body makes thousands of them. They work as signaling molecules: they bind to receptors on cell surfaces and kick off intracellular cascades that handle everything from immune responses to tissue repair. Short, targeted chemical messages between cells.
What makes therapeutic peptides different from the broader category is specificity. A broad-spectrum anti-inflammatory drug dampens immune function everywhere. A peptide can, in theory, activate one repair pathway in one type of tissue. That specificity is why over 80 peptide drugs have received FDA approval across conditions from diabetes to cancer.
BPC-157 and thymosin beta-4 are not in that group. They're not FDA-approved drugs. They exist in a regulatory gray zone, and the distinction between "approved peptide therapeutic" and "peptide you bought from a website" is not trivial.
Your body already relies on peptide signaling for collagen synthesis, wound closure, and blood vessel formation. The compounds discussed here are either naturally occurring peptides that have been isolated and synthesized, or fragments of larger endogenous proteins.
BPC-157: the gut peptide that blew up online
Body Protection Compound-157 is a chain of 15 amino acids first isolated from human gastric juice by Dr. Predrag Sikiric in 1993. Naturally, it helps maintain the stomach lining. Nobody paid much attention until researchers noticed it doing things far outside the gut.
In preclinical animal models, BPC-157 has shown regenerative effects in tendons, ligaments, muscles, bones, nerves, corneas, and blood vessel linings. That's an unusual range for a single compound, and it's a big part of why interest exploded online.
How it works at the molecular level
A 2025 systematic review covering 36 studies from 1993 to 2024 mapped out several overlapping mechanisms. BPC-157 stimulates VEGF (vascular endothelial growth factor), promoting new blood vessel formation. It activates the ERK1/2 signaling cascade, which drives cell growth and migration. It boosts nitric oxide production through the Akt-eNOS pathway, widening blood vessels and improving blood flow to injured tissue. And it dials down inflammatory markers like IL-6, TNF-alpha, and cyclooxygenase-2.
It seems to speed up healing by getting more blood to damaged tissue while turning down inflammation and ramping up growth signals. At least in animals.
What the animal data shows
The preclinical evidence is extensive for a compound that's never been through a proper clinical trial. In rats with severed Achilles tendons, BPC-157 improved structural and biomechanical outcomes. In quadriceps crush injuries, it restored muscle structure and motor function. Rabbits with nonunion fractures showed bone healing comparable to autologous bone marrow injections, which is a standard clinical treatment.
| Tissue type | Animal model | What happened |
|---|---|---|
| Achilles tendon | Rat transection | Improved biomechanical strength, less inflammation |
| Quadriceps muscle | Rat transection/crush | Restored muscle structure and motor function |
| Medial collateral ligament | Rat transection | Reduced valgus instability, restored biomechanics |
| Bone nonunion | Rabbit | Callous mineralization, lamellar bone formation |
| Inflammatory arthritis | Rat adjuvant model | Less paw inflammation and stiffness |
BPC-157 gets metabolized in the liver, has a half-life under 30 minutes, and clears through the kidneys. Safety studies in animals found no adverse effects across several organ systems, no mutagenicity in the Ames test, and no genotoxic or teratogenic effects. Researchers couldn't find a minimum toxic dose or lethal dose. Which sounds reassuring until you remember that these were all animal studies.
Thymosin beta-4: repair and recovery at the cellular level
Thymosin beta-4 (Tβ4) is a 43-amino-acid peptide found throughout the body, with high concentrations in the thymus, spleen, platelets, and white blood cells. It makes up 70-80% of all beta-thymosin in the human body. TB-500, the name you'll see on vendor sites, is a synthetic fragment of the full protein.
Unlike BPC-157, which came out of gastric biology, thymosin beta-4 is rooted in immunology and wound healing. When tissue gets damaged, platelets and macrophages release it as part of the immediate damage-control response. It's one of the body's built-in repair dispatchers.
What it does biologically
A review in Frontiers in Endocrinology broke down how thymosin beta-4's activity maps to specific stretches of its amino acid sequence. The first four amino acids handle anti-inflammatory and antifibrotic effects. Amino acids 1-15 block apoptosis (programmed cell death). Amino acids 17-23 trigger angiogenesis and hair follicle growth.
So a single 43-amino-acid peptide promotes blood vessel formation via VEGF upregulation, cuts scar tissue by reducing myofibroblasts, blocks inflammatory cascades through NF-κB inhibition, and protects cells from oxidative damage. That's a lot of activity for one molecule.
In wound healing studies, topical or intraperitoneal thymosin beta-4 accelerated reepithelialization by 42% at day 4 and 61% at day 7 versus saline controls. It also worked in diabetic and aged mice, where healing is notoriously slow.
Cardiac and eye research
The strongest thymosin beta-4 data comes from cardiac repair studies. Phase I trials in healthy volunteers found no dose-limiting toxicities. A Phase II trial in heart attack patients showed reduced scar volume. A randomized double-blind trial tested it during congenital heart surgery in children, and it improved ischemia-reperfusion outcomes.
There's also Phase II trial data for dry eye syndrome (35.1% reduction in ocular discomfort) and chronic ulcers (healing accelerated by nearly a month). Of the two peptides in this article, thymosin beta-4 has a much more developed clinical track record, though it's still not FDA-approved for anything. For comparison, GLP-1 receptor agonists went through a decade-plus pipeline before reaching the market.
| Application | Study type | Result |
|---|---|---|
| Wound healing | Preclinical (mice) | 42-61% faster reepithelialization vs controls |
| Dry eye syndrome | Phase II clinical trial | 35.1% reduction in ocular discomfort |
| Acute MI | Phase II clinical trial | Reduced cardiac scar volume |
| Congenital heart surgery | Randomized double-blind trial | Improved ischemia-reperfusion outcomes |
| Stasis/pressure ulcers | Phase II clinical trial | Healing accelerated by nearly a month |
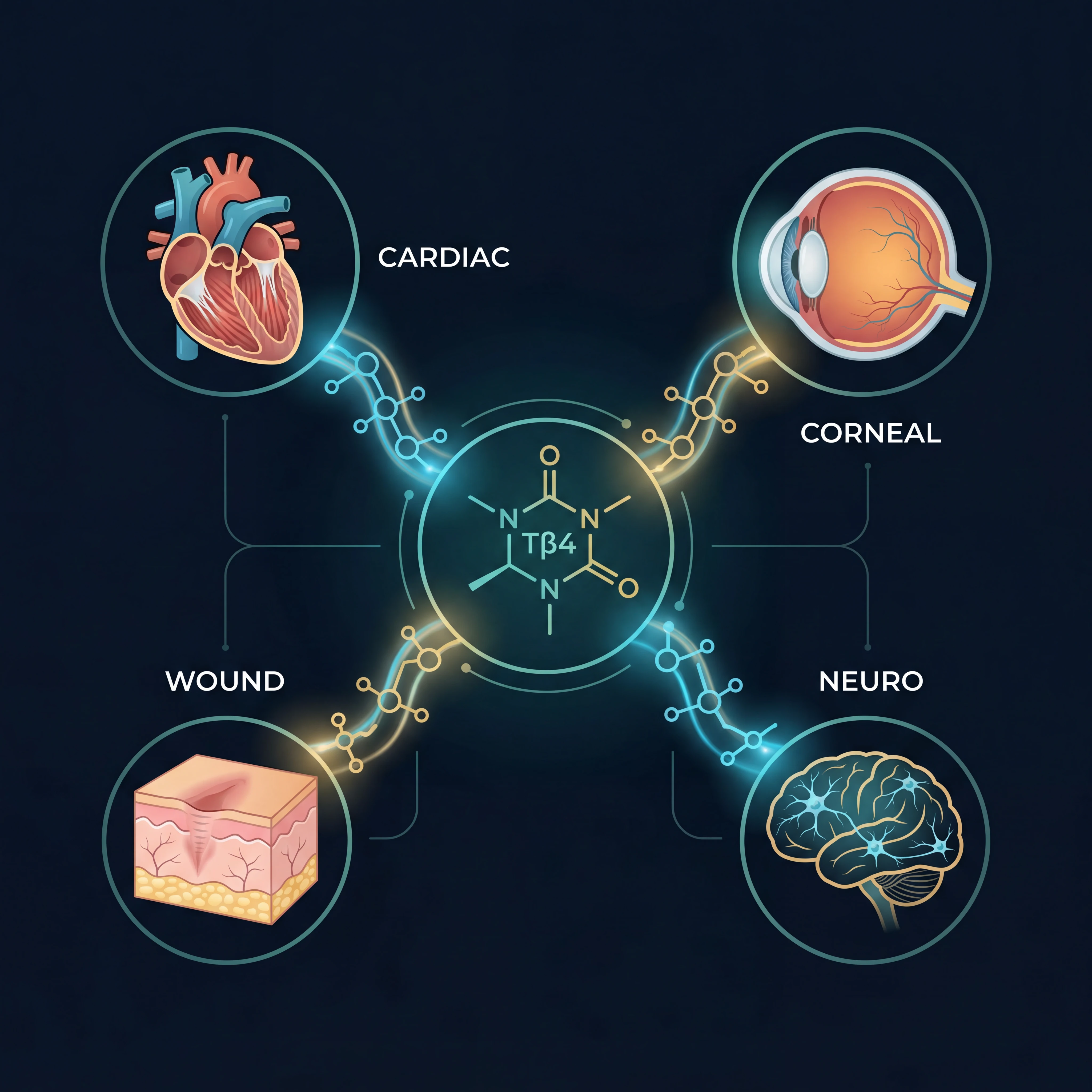
How BPC-157 and thymosin beta-4 actually compare
These two get lumped together constantly in online discussions. They shouldn't be. They're different compounds, at different stages of research, in different regulatory positions.
| Feature | BPC-157 | Thymosin beta-4 |
|---|---|---|
| Origin | Human gastric juice | Thymus gland, platelets, white blood cells |
| Size | 15 amino acids | 43 amino acids |
| Primary mechanism | VEGF, ERK1/2, nitric oxide | PI3K/Akt/eNOS, Notch, Wnt pathways |
| Human clinical trials | 3 pilot studies only | Multiple Phase I and Phase II trials |
| FDA status | Category 2 bulk drug (can't be compounded) | Not approved; investigational |
| WADA status | Specifically banned (2022) | Banned under peptide hormone category |
| Half-life | <30 minutes | Variable by route |
| Research focus | Musculoskeletal, GI | Cardiac, wound healing, ophthalmology |
They share some biology: both promote angiogenesis through VEGF-related pathways, both reduce inflammatory cytokines, both show broad tissue-protective effects in preclinical models. But thymosin beta-4 has actual Phase II trial data in humans for specific conditions. BPC-157 has three pilot studies with fewer than 30 total participants and no placebo controls.
When someone on a forum stacks these as equivalent recovery tools, they're treating a compound with genuine (if limited) clinical evidence the same as one that's almost entirely preclinical. That's like comparing a drug in Phase II trials to one that hasn't even filed an IND application.
The legal situation (it's complicated)
The regulatory picture here isn't a simple legal-or-not binary. There are at least three separate regulatory frameworks that apply, and they don't all agree.
BPC-157's Category 2 classification
In September 2023, the FDA classified BPC-157 as a Category 2 bulk drug substance. That means it raises significant safety concerns. Licensed pharmacies and outsourcing facilities cannot legally use it in compounded medications under sections 503A or 503B of the Federal Food, Drug, and Cosmetic Act. In December 2024, the FDA's Pharmacy Compounding Advisory Committee raised further concerns about immunogenicity risks with compounded peptides.
The FDA has explicitly stated it would consider regulatory action against any compounder using BPC-157.
What's not illegal
BPC-157 is not a DEA-scheduled substance. Possessing it is not a criminal offense the way possessing anabolic steroids without a prescription would be. Products labeled "research chemicals" or "not for human consumption" continue to be sold online. The practical result: you can buy it, but no regulated medical professional can legally compound or prescribe it for you.
Sports bans
WADA specifically banned BPC-157 by name in 2022 under its S0 Unapproved Substances category. The NFL and UFC have specific prohibitions. The NCAA, MLB, PGA, and other organizations ban it under broader peptide hormone categories. If you compete in anything with drug testing, using either peptide puts your eligibility at risk.
What actually goes wrong with unregulated peptides
The biggest safety concern with BPC-157 isn't necessarily the compound itself. Based on animal data, it appears well-tolerated at even very high doses. The problem is what you're actually injecting when you buy from an unregulated source.
Products sold outside FDA oversight have no quality assurance for potency, purity, or sterility. A 2025 narrative review flagged several theoretical risks based on BPC-157's mechanisms: pathologic angiogenesis (potentially feeding tumor growth), toxic metabolite formation from proline metabolism, and nitric oxide overproduction that could mess with hemoglobin function and how your body processes other drugs.
The same review also found that BPC-157 appears to modulate angiogenesis selectively based on tissue context. It actually showed anti-tumor effects in some animal models. And preclinical safety evaluations couldn't establish a toxic dose at all.
The more concrete dangers are contamination and mislabeling. Without regulated manufacturing, a vial labeled "BPC-157" might contain incorrect doses, degraded peptides, bacterial endotoxins, or substances that aren't listed at all. Injectable products are the biggest concern because needles bypass every natural barrier your body has against contaminants. (This is the same reason the FDA cracked down on compounded GLP-1 versions containing B12, too.)
If recovery is the goal, it's worth knowing that cold exposure therapy and strategies for supporting your body's own antioxidant systems have much stronger human evidence behind them.
What we know from human studies so far
Animal studies fail to translate into human benefit about 90% of the time across drug development. The preclinical data on both peptides is interesting, but interesting and proven are different things.
BPC-157 in humans
As of early 2026, three human studies on BPC-157 have been published.
- Chronic knee pain (Lee and Padgett, 2021): Twelve patients got intraarticular BPC-157 injections. Seven reported feeling better for over six months. No control group, no blinding, no way to separate placebo from real effect.
- Interstitial cystitis (Lee et al., 2024): Intravesicular administration in a small group. No side effects reported. Participants were screened for fever, rash, nausea, and worsening symptoms. None turned up.
- IV safety pilot (Lee and Burgess, 2025): Two healthy adults received intravenous BPC-157. It was well tolerated. Plasma levels returned to baseline within 24 hours.
Fewer than 30 total participants across all three studies. No placebo controls. No efficacy data beyond self-reports. This isn't evidence that BPC-157 doesn't work. It's evidence that nobody has properly tested whether it does.
Thymosin beta-4 in humans
Thymosin beta-4 has a substantially stronger clinical record. Phase I trials confirmed safety with no dose-limiting toxicities. Phase II data shows measurable benefit in dry eye syndrome (35.1% less ocular discomfort), heart attack recovery (reduced scar volume), and chronic wound healing (ulcers healed nearly a month faster).
A randomized double-blind trial during pediatric heart surgery also showed benefit for ischemia-reperfusion injury. That's real clinical evidence, though still preliminary by FDA-approval standards.
For perspective, creatine's benefits beyond muscle took decades of accumulating human data to establish. Peptide therapeutics are early in that process.

Frequently Asked Questions
Is BPC-157 legal to buy in the United States?
It's not a DEA-scheduled substance, so buying it isn't a criminal offense. But the FDA classified it as a Category 2 bulk drug substance in 2023, which means pharmacies and outsourcing facilities can't compound it. The products you see online sold as "research chemicals" or "dietary supplements" aren't subject to FDA quality or safety oversight. You can buy them, but nobody is checking what's actually in them.
Can my doctor prescribe BPC-157 or thymosin beta-4?
Neither has FDA approval for any medical use. BPC-157 can't be legally compounded under current rules. Thymosin beta-4 is still investigational. Some cash-pay clinics offer them, but there's no regulatory quality assurance and no established dosing guidelines for human use. Your doctor won't find either in any prescribing database.
Are these peptides banned in sports?
Yes. WADA banned BPC-157 by name in 2022. The NFL and UFC have specific prohibitions. The NCAA, MLB, and PGA ban it under broader peptide hormone categories. Thymosin beta-4 falls under similar bans. Testing positive for either means sanctions, regardless of how you got it.
What are the main safety concerns with peptides from online sources?
Contamination, wrong doses, and degradation. There's no guarantee that what's in the vial matches what's on the label, that it's sterile, or that it hasn't broken down during shipping. Injectables are the biggest worry because a needle bypasses every natural barrier your body has. There's also no chronic dosing safety data in humans for either compound.
Will BPC-157 or thymosin beta-4 ever get FDA approval?
Thymosin beta-4 has a more realistic path since it already has Phase II data in ophthalmology and cardiology. BPC-157 would need to start from zero with formal clinical trials, a process that costs hundreds of millions of dollars and typically takes 10-15 years. Researchers have been calling for proper human trials, but as of now, none are registered in major clinical trial databases for BPC-157.
Related Articles
- Collagen Supplements -- Do They Actually Work for Skin, Joints, and Gut? Collagen peptides are among the few peptide supplements with substantial human clinical data.
- GLP-1 Weight Loss Drugs: Safety, Side Effects, and Natural Alternatives GLP-1 agonists are the most commercially successful peptide drug class and offer a useful comparison point.
- Creatine Benefits Beyond Muscle -- Brain, Bone, and Longevity A supplement with decades of human research showing how long it takes to build a strong evidence base.
- Glutathione: The Master Antioxidant Your Body Makes (and How to Boost It) Endogenous antioxidant support with better-established human evidence for recovery and cellular protection.
- Cold Plunge and Ice Bath Benefits: What Happens to Your Body Evidence-based recovery strategies with stronger clinical backing than experimental peptides.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.










