Hormone Replacement Therapy (HRT) — Benefits, Risks, and Modern Evidence
Discover current evidence on hormone replacement therapy for menopause, including benefits, risks, timing, and how to choose the right type for you.
12 Min Read
What Hormone Replacement Therapy Actually Does
Hormone replacement therapy replaces estrogen and, when needed, progesterone that the ovaries stop producing during menopause. That drop triggers the symptoms most women know too well: hot flashes, night sweats, vaginal dryness, mood shifts, broken sleep, and thinning bones.
About 75% of menopausal women experience symptoms, and roughly a third say those symptoms are severe enough to get in the way of daily life. Despite that, only about 10% of eligible women in the UK currently take HRT. That gap has more to do with decades of fear than with the actual evidence.
HRT comes in two main forms. Estrogen therapy (ET) is prescribed alone for women who have had a hysterectomy. Estrogen-progestogen therapy (EPT) combines both hormones and is necessary for women with an intact uterus, because unopposed estrogen can stimulate the uterine lining and raise endometrial cancer risk.
The goal: restore hormone levels enough to relieve symptoms and protect bone density without introducing unnecessary risk. How well that balance is struck depends on the type of HRT, the dose, the delivery method, and when you start it.
The WHI Study Changed Everything, Then We Misread It
In 2002, the Women's Health Initiative (WHI) trial made international headlines when researchers halted the estrogen-plus-progestin arm early. The results appeared to show that HRT increased the risk of breast cancer, heart disease, and stroke. Millions of women stopped their prescriptions almost overnight, and many doctors stopped prescribing HRT entirely.
The problem? The study's participants averaged 63 years old, well past the typical age of menopause. Many had pre-existing cardiovascular risk factors. The findings were valid for that population, but they were overgeneralized to all women at all ages and all formulations.
Reanalysis of the WHI data tells a different story. Among women aged 50-59 who started HRT near menopause onset, there was no increased risk of coronary heart disease. The estrogen-only arm actually showed fewer breast cancers and lower mortality over long-term follow-up. In July 2025, an FDA expert panel formally acknowledged that early interpretations of the WHI data had been applied too broadly.
The key takeaway: The WHI didn't prove that HRT is dangerous for menopausal women. It proved that starting hormones in your 60s or 70s, years after menopause, carries risks that don't apply to women who start earlier.
That distinction matters. It's the reason modern guidelines now emphasize timing, dosage, and delivery route rather than blanket warnings.
What 70 Years of Research Show About the Benefits
The evidence supporting HRT for the right candidates is stronger than many people realize.
Vasomotor symptom relief. Estrogen therapy reduces hot flash frequency by 70-80% compared to baseline, with the North American Menopause Society estimating a 75% reduction versus placebo. Most women feel relief within 2-4 weeks, with full effect by 8-12 weeks. Nothing else comes close for hot flashes and night sweats.
Bone protection. HRT remains one of the most effective ways to prevent postmenopausal osteoporosis. Estrogen directly slows bone breakdown, and studies consistently show reduced fracture risk at the hip, spine, and wrist during HRT use. For women also interested in bone-protective nutrients like vitamin D, HRT works through a different mechanism entirely.
Metabolic benefits. HRT reduces new-onset type 2 diabetes by 20-30%, a finding that holds up across multiple randomized controlled trials and meta-analyses. It also improves insulin sensitivity and has a favorable effect on body composition.
Genitourinary symptom relief. Between 50-70% of postmenopausal women develop genitourinary syndrome of menopause (GSM), which includes vaginal dryness, urinary urgency, recurrent infections, and pain during intercourse. Local vaginal estrogen works well and carries minimal systemic absorption. Even women on systemic HRT may need additional local treatment, since about one in three still report GSM symptoms.
Cardiovascular protection (timing-dependent). For women who begin HRT before age 60 or within 10 years of menopause, the evidence points to reduced coronary heart disease and lower all-cause mortality. This protection appears strongest with estrogen-only therapy but extends to combined formulations when started within the timing window. Estrogen improves vascular function, reduces arterial stiffness, and shifts cholesterol profiles in a favorable direction. Those effects protect healthy blood vessels but may destabilize plaques that have already built up in older women.
| Benefit | Evidence Strength | Key Finding |
|---|---|---|
| Hot flash reduction | Strong (RCTs) | 70-80% reduction vs. baseline |
| Bone fracture prevention | Strong (RCTs) | Reduced hip, spine, wrist fractures |
| Diabetes prevention | Strong (RCTs, meta-analyses) | 20-30% reduction in new-onset T2D |
| Genitourinary relief | Strong (RCTs) | Effective for GSM in 50-70% of women |
| Cardiovascular protection | Moderate (age-dependent) | Benefit when started before age 60 |
| Mood improvement | Moderate | Reduces depressive symptoms in perimenopause |
How Real Are the Risks?
The risks are real, but they're smaller and more specific than most women have been led to believe. The same therapy that poses minimal risk for a 52-year-old may not be appropriate for a 68-year-old.
Breast cancer. This is the risk that scares women most, and it deserves careful parsing. Estrogen-only HRT shows no greater risk of breast cancer than not taking hormones. The WHI estrogen-only arm actually showed fewer breast cancers over long-term follow-up. The increased risk comes from combined estrogen-plus-synthetic-progestogen therapy and depends on duration: for every 1,000 women starting HRT at age 50 and using it for 10 years, about 6 extra cases of breast cancer occur. That's comparable to drinking 1-2 glasses of wine per night or being overweight.
The type of progestogen also matters. A systematic review and meta-analysis found that micronized progesterone carries lower breast cancer risk than synthetic progestins like medroxyprogesterone acetate (MPA). When transdermal estrogen is combined with micronized progesterone, no increased breast cancer risk shows up for the first five years of use.
Blood clots (VTE). Oral estrogen raises the risk of venous thromboembolism. Transdermal estrogen (patches, gels, sprays) does not. This is one of the clearest safety distinctions in HRT, and it's a major reason most current guidelines favor transdermal delivery, especially for women with elevated clot risk.
Stroke. A large Korean cohort study of over 550,000 women found that current oral HRT use was linked to higher ischemic stroke risk, but a Swedish cohort showed no increased stroke risk when HRT was started within 5 years of menopause, regardless of route. Overall, HRT-associated risks of breast cancer, stroke, and VTE are rare, at fewer than 10 events per 10,000 women per year.
Women looking into magnesium supplementation for sleep and mood or other supportive approaches should weigh these risk numbers against their own symptom burden and risk profile.
| Risk Factor | Absolute Risk Increase | Context |
|---|---|---|
| Breast cancer (E+P) | ~6 extra cases per 1,000 over 10 years | Similar to 1-2 drinks/day or obesity |
| Breast cancer (E-only) | No increase (possible decrease) | WHI showed fewer cases long-term |
| VTE (oral) | Increased | Transdermal route eliminates this risk |
| VTE (transdermal) | No increase | Preferred for women with clot risk |
| Stroke | <10 events per 10,000 women/year | Timing-dependent; lower when started early |
| Ovarian cancer | ~1 extra case per 1,000 over 5 years | Small absolute increase |
Comparing HRT Types: Which Formulation Fits?
Not all HRT is the same. The type of hormone, the dose, and how it gets into your body make a real difference in both effectiveness and safety.
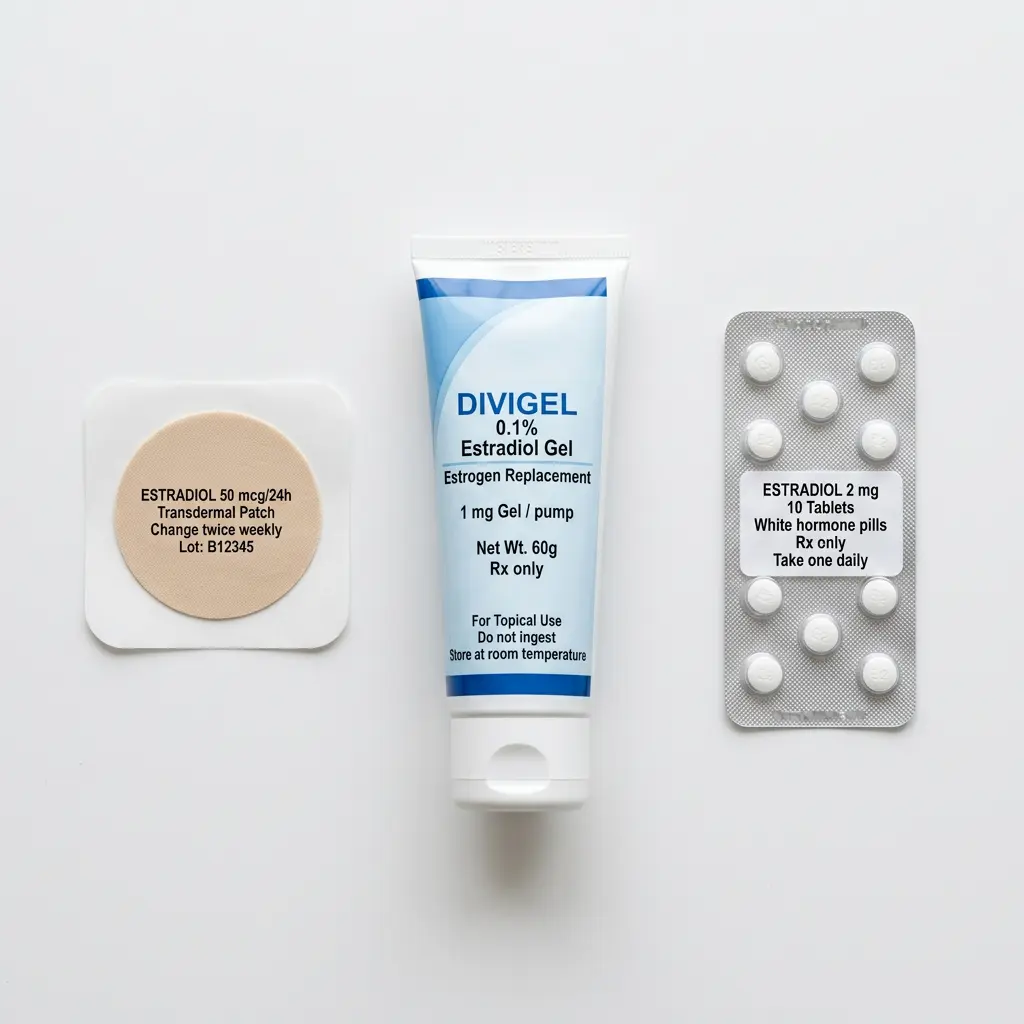
Estrogen types. Conjugated equine estrogens (CEE), derived from pregnant mare urine, were the formulation used in the WHI trial. Micronized 17-beta estradiol, which is bioidentical to the estrogen human ovaries produce, is now preferred by most guidelines. Estradiol shows more favorable outcomes than CEE across cardiovascular and metabolic markers.
Progestogen types. This is where the safety differences get stark. Synthetic progestins like MPA and norethisterone carry higher breast cancer risk with prolonged use. Micronized progesterone (marketed as Prometrium or Utrogestan) has a better safety profile across VTE, breast cancer, and cardiovascular outcomes. Dydrogesterone also shows favorable results.
| Dose Level | Conjugated Estrogens (oral) | Micronized Estradiol (oral) | Estradiol Patch |
|---|---|---|---|
| Ultra-low | 0.3 mg/day | 0.25 mg/day | 14 mcg/day |
| Low | 0.45 mg/day | 0.5 mg/day | 25 mcg/day |
| Standard | 0.625 mg/day | 1.0 mg/day | 37.5-50 mcg/day |
Delivery methods. Estrogen can be taken as a pill, patch, gel, spray, vaginal cream, ring, or suppository. Transdermal delivery (patches and gels) bypasses the liver, which eliminates the clotting risk that comes with oral estrogen. NAMS, EMAS, ACOG, and the British Menopause Society all recommend transdermal estrogen for women with elevated VTE risk, migraines with aura, gallbladder disease, diabetes, or obesity.
Compounded "bioidentical" hormones. Between 1 and 2.5 million American women over 40 use compounded bioidentical hormones, often marketed as "natural" or "custom-made." But compounded preparations are not FDA-approved, not tested for safety or effectiveness, and don't have consistent quality control. Compounding pharmacies aren't required to report side effects to the FDA.
Here's the distinction that gets lost in the marketing: FDA-approved bioidentical hormones (like micronized estradiol and micronized progesterone) come from the same plant sources and have the same molecular structure as compounded versions, but they're rigorously tested and manufactured under strict quality standards. The American College of Obstetricians and Gynecologists explicitly recommends against compounded bioidentical HRT when FDA-approved alternatives exist. Salivary hormone testing, which is often used to guide compounded dosing, has never been validated through rigorous research.
Emerging options. Estetrol (E4), a natural estrogen previously found only during pregnancy, is being studied for menopausal use. Early Phase II data suggests it may have a thrombotic safety profile closer to progestin-only pills while still providing effective symptom relief at 15 mg daily.
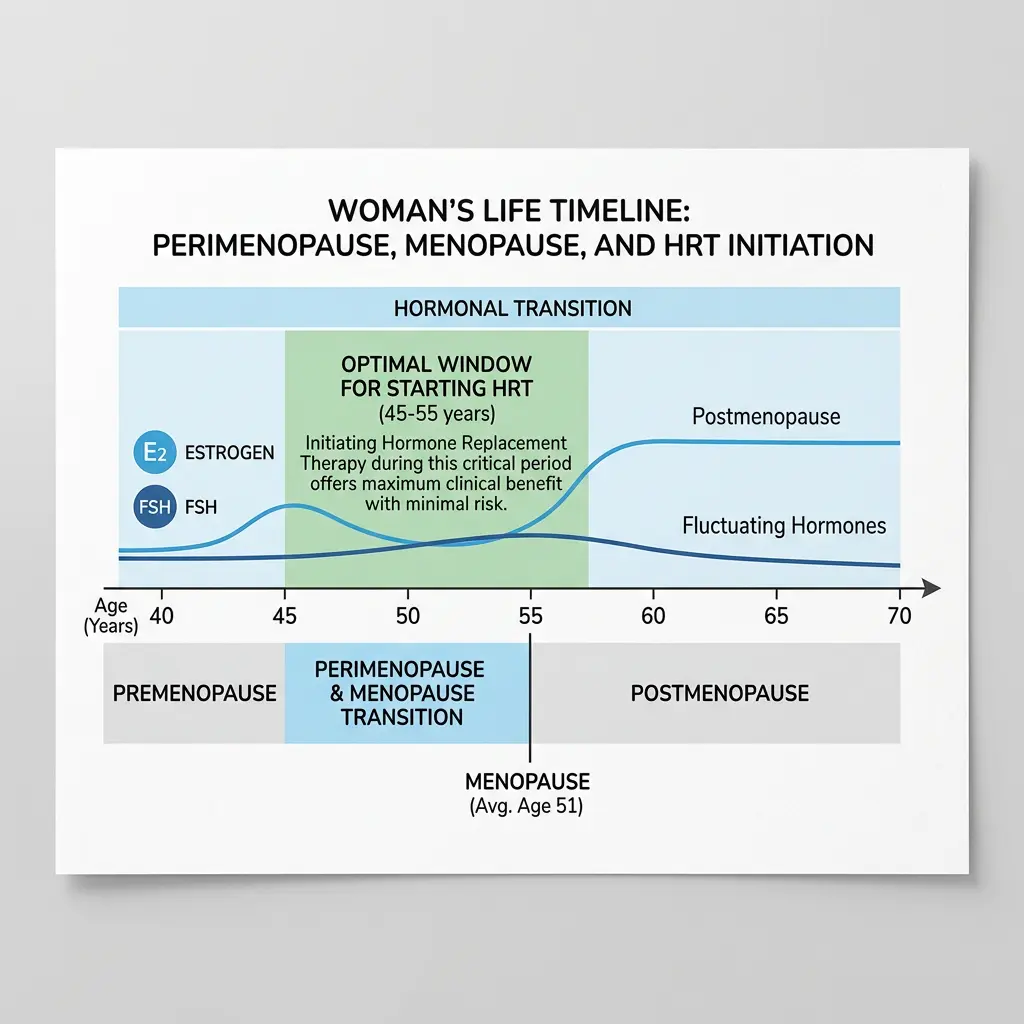
The Timing Window That Changes Everything
If one concept has reshaped modern HRT prescribing, it's the "timing hypothesis," also called the "window of opportunity." The idea is simple: the benefits and risks of HRT depend heavily on when you start relative to menopause onset.
A systematic review and meta-regression analysis confirmed that women who begin HRT within 10 years of menopause or before age 60 see reduced all-cause mortality and cardiovascular benefit. Women who start more than 10 years after menopause or after age 60 face increased cardiovascular and stroke risk.
WHI age-stratified reanalysis from 2024 backed this up: women aged 50-59 with moderate-to-severe vasomotor symptoms got relief without increased cardiovascular risk. The data was clear enough that the 2025 Korean Menopausal Hormone Therapy Guidelines, along with NAMS and EMAS, all built the timing window into their formal recommendations.
For women already past that window, starting HRT is generally not recommended for cardiovascular protection, though it may still be considered for severe vasomotor symptoms on a case-by-case basis. No major medical society recommends starting HRT at age 70 or older.
All four major guideline bodies, NAMS, EMAS, ACOG, and the British Menopause Society, now recommend using the lowest effective dose for the shortest necessary duration, with regular reassessment. But "shortest necessary duration" no longer means a rigid five-year limit. Continuing past age 65 is considered appropriate for women who still benefit and whose risk profiles remain favorable. That's a big change from the post-WHI era, when arbitrary time limits pushed many women off treatment they still needed.
Bottom line: if you're in perimenopause or early menopause and considering HRT, the evidence favors starting sooner rather than waiting. Women looking into supportive strategies, whether collagen supplements for joint and skin health or evidence-based supplements for women over 40, may find these helpful alongside HRT, but they won't replace what hormones do for hot flashes and night sweats.
Myths vs. Facts: Separating Fear From Evidence
| Myth | What the Evidence Actually Shows |
|---|---|
| HRT causes breast cancer | Estrogen-only HRT does not increase breast cancer risk. Combined therapy shows a small, duration-dependent increase comparable to lifestyle factors like alcohol use. |
| HRT causes heart attacks | When started within 10 years of menopause or before age 60, HRT reduces cardiovascular risk. The WHI's cardiac findings applied to women who started in their 60s. |
| All HRT is the same | Formulation, dose, and delivery route matter a lot. Transdermal estradiol plus micronized progesterone has a very different risk profile than oral conjugated estrogen plus MPA. |
| Compounded bioidenticals are safer than "synthetic" hormones | FDA-approved bioidentical hormones are molecularly identical and rigorously tested. Compounded products lack quality control, consistent dosing, and safety monitoring. |
| You should take HRT for the shortest time possible | Major societies now say continuing beyond age 65 is acceptable for selected women with periodic reassessment. Duration should be individualized, not set by an arbitrary cutoff. |
| "Natural" menopause treatments eliminate the need for HRT | Lifestyle changes, supplements, and complementary therapies can help with mild symptoms. For moderate-to-severe hot flashes, nothing matches HRT's 70-80% reduction rate. |
Frequently Asked Questions
How long can you safely stay on hormone replacement therapy?
There is no fixed cutoff. Current guidelines from NAMS, EMAS, ACOG, and the British Menopause Society recommend using the lowest effective dose and reassessing regularly. Continuing beyond age 65 is considered acceptable for women who still benefit, as long as their doctor reviews their risk profile periodically. The old "five-year rule" is no longer supported by the evidence.
Is transdermal HRT safer than pills?
For certain risks, yes. Transdermal estrogen (patches, gels, sprays) bypasses the liver's first-pass metabolism, which eliminates the increased risk of blood clots and may carry a lower stroke risk compared to oral formulations. Most current guidelines recommend transdermal delivery for women with elevated clot risk, obesity, migraines with aura, or gallbladder disease.
What is the difference between bioidentical and synthetic hormones?
Bioidentical hormones have the same molecular structure as hormones the body naturally produces. Synthetic hormones have a different chemical structure. Both can be effective, but bioidentical estradiol and micronized progesterone show more favorable safety profiles in some studies. The more important distinction is between FDA-approved bioidenticals (regulated, tested, consistent) and compounded bioidenticals (unregulated, untested, inconsistent dosing).
Can HRT help with brain fog and mood changes during menopause?
Estrogen receptors are abundant in the brain, and estrogen plays a role in neurotransmitter function, blood flow, and neuroprotection. Research shows that HRT can improve depressive symptoms during perimenopause and may support cognitive function when started during the timing window. That said, HRT is not currently recommended solely for cognitive protection or dementia prevention.
Does HRT cause weight gain?
The evidence doesn't support this common worry. Menopause itself shifts body composition, increasing abdominal fat and decreasing lean muscle mass, whether or not women take HRT. Some studies suggest HRT actually helps maintain a better body composition by reducing central fat accumulation. The hormonal changes of menopause, not the treatment, drive weight redistribution. Managing chronic stress and cortisol levels also plays a role in menopausal weight patterns.
Related Articles
- Collagen Supplements: Do They Actually Work for Skin, Joints, and Gut? — What the evidence says about collagen peptides for joint pain, skin elasticity, and gut integrity during aging.
- Magnesium Types Explained: Glycinate, Citrate, Threonate — Which form of magnesium works best for sleep, muscle cramps, mood, and cognitive function.
- Weight Loss Supplements for Women Over 40 — Research-backed supplements that may support metabolic health during and after the menopausal transition.
- The Hidden Toll of Low-Level Stress: How Chronic Tension Wrecks Your Hormones — How chronic cortisol elevation interacts with reproductive hormones and strategies for HPA axis recovery.
- Vitamin D Benefits, Deficiency Signs, and Safe Dosing — Essential information on a nutrient that works alongside HRT to protect bone density and immune function.
Medical Disclaimer
This article is for informational and educational purposes only and is not medical advice, diagnosis, or treatment. Always consult a licensed physician or qualified healthcare professional regarding any medical concerns. Never ignore professional medical advice or delay seeking care because of something you read on this site. If you think you have a medical emergency, call 911 immediately.










